ABC World News – Integrated Medicine
July 20, 2015
PALS Certification Kingwood
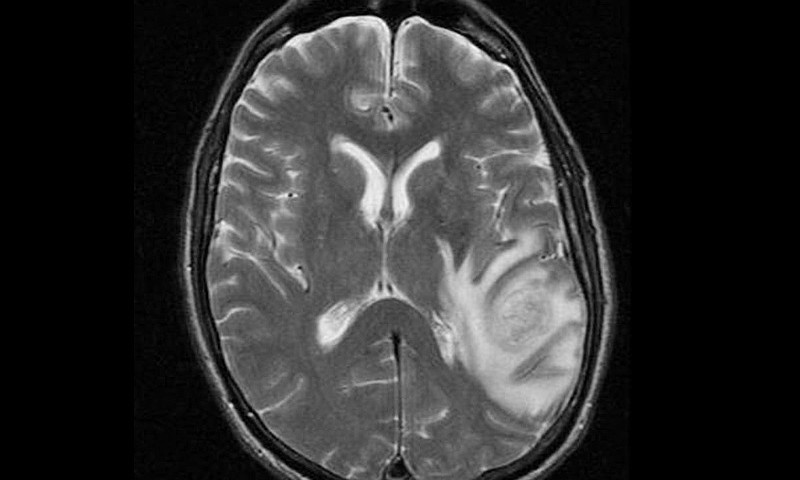
July 27, 2015The use of new optical coherence tomography (OCT) technology may more accurately distinguish between brain tumor cells and healthy brain tissue, enabling more thorough removal of brain tumors without damaging normal brain cells, a new study suggests.
“This new technology can distinguish what is cancer and what is normal brain tissue better than anything else we have. We are finally cracking the code,” lead investigator Alfredo Quinones-Hinojosa, MD, professor of neurosurgery, neuroscience and oncology at the Johns Hopkins University School of Medicine, Baltimore, Maryland, told Medscape Medical News.
“As a neurosurgeon, I’m in agony when I’m taking out a tumor. If I take out too little, the cancer could come back; too much, and the patient can be permanently disabled. We think optical coherence tomography has strong potential for helping surgeons know exactly where to cut,” he said.
With current technology it is difficult to distinguish between cancer cells and normal brain cells, particularly at the edge of the tumor, he added.
“It is not like in other areas on the body where extra tissue can be taken to be on the safe side. You can’t do this in the brain. We obviously do not want to remove healthy brain cells, as this could compromise speech, motor and cognitive function. So we end up leaving parts of the tumor in. With OCT we can be much more accurate.”
“It has recently been shown that 70% of a brain tumor needs to be removed to have an effect on survival, and survival improves with every extra 5% of tumor removed. Our results suggest this new technology should enable up to 95% of the tumor to be removed, which could have dramatic effects on survival,” lead author and medical student, Carmen Kut, told Medscape Medical News.
The technology was developed by a group led by Xingde Li, PhD, professor of biomedical engineering at Johns Hopkins. He explained that the OCT technology provides direct visual guidance to the surgeon in the intraoperative setting. “It works like ultrasound but uses light instead of sound to give a picture of the tissue.”
“At present a preoperative MRI shows the position of the tumor but the brain shifts during surgery so there is some uncertainty about the exact limits of the cancerous tissue. Microscopes can also be used during surgery but these only show the surface appearance of the tissue.
“The OCT technology is used in real time during the surgery — by pointing the probe at each individual piece of tissue it detects which tissue is cancerous and needs to be removed and which is normal so can be left in. The results are shown in a real time coloured map, with red signaling cancer and green signaling healthy cells,” he added.
In the current paper, published June 17 in Science Translational Medicine, the researchers imaged multiple brain tumor tissue samples fresh from surgery from 32 patients with brain cancer. The OCT technology was used to compare how light passes through the cancer cells compared to healthy cells.
From these findings they calculated the optical attenuation coefficient — a measure of how much light from the probe is absorbed or reflected in a particular tissue — and how this differs for cancer cells and healthy cells.
Kut explained that in healthy white matter the myelin sheath bounces light back, giving a high optical attenuation. But when cancer invades brain cells it causes the myelin in the white matter to break down and the optical attenuation will drop. This is unique to brain cancer.
In this study the researchers established that OCT achieved high sensitivity and specificity for brain cancer tissue at an attenuation threshold of 5.5 mm–1. Optical attenuation values below 5.5 mm–1 signify cancer cells, and values above this level signify healthy tissue.
“At this threshold, OCT has a sensitivity of detecting cancer cells of 92% to 100%. And it has a specificity (identifying healthy tissue) of 80% to 100%,” Kut said.
This was then validated in an in vivo study in live mice implanted with human brain tumor cells.
The current results lay the foundations for clinical trials, beginning with a phase 1 study to start later this summer.
Sci Transl Med. Published online June 17, 2015. Abstract